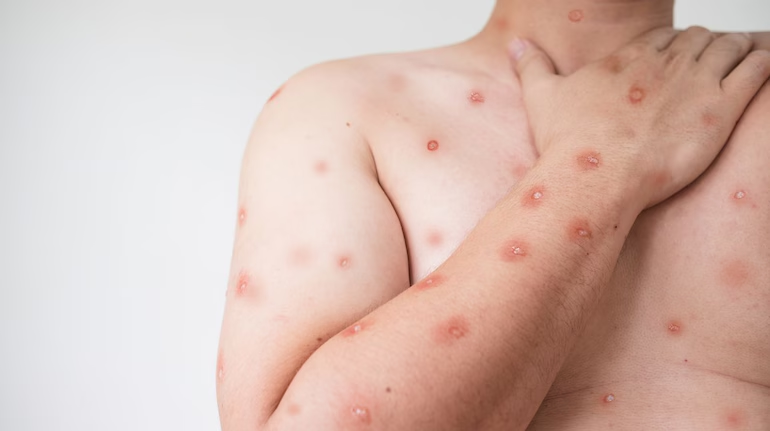
New Delhi: The Centre on Thursday asked all states to identify facilities and trained human resources for taking care of both suspected and confirmed mpox cases after India reported its first case of the strain that had last month prompted WHO to declare a public health emergency. In a letter to states and Union territories, Union Health Secretary Apurva Chandra said samples from skin lesions of any patient with suspected mpox symptoms should be immediately sent to designated labs.
Samples testing positive will be sent to the Indian Council of Medical Research-National Institute of Virology (ICMR-NIV) for genome sequencing to determine the clade.
Chandra said the World Health Organization (WHO) on August 14 announced that the current outbreak of mpox (previously known as Monkeypox) disease was a Public Health Emergency of International Concern (PHEIC).
This is the second time such mpox disease-associated PHEIC has been declared by WHO under the International Health Regulations, 2005, to which India is a signatory.
The previous outbreak in 2022 was caused by the Clade 2 strain of the virus, the letter said.
“The 2024 PHEIC is related to mpox virus Clade 1, which is more virulent and more transmissible than mpox Clade 2. This clade has been found outside Democratic Republic of Congo (DRC) only during the current outbreak. Outside Africa, one case each of mpox Clade 1b has been recently reported from Sweden and Thailand. India is thus the third non-African country to report a case of Clade 1b mpox infection recently,” Chandra said.
According to available information, clinical presentation of mpox Clade 1 in adults was similar to Clade 2. However, rate of complications may be higher in Clade 1 than with Clade 2 infections.
Chandra listed some public health actions required to be undertaken to prevent/minimise the risk of further spread of mpox in India as part of which all states and Union territories have been urged to undertake appropriate activities to make communities aware about the disease, its modes of spread, need/importance of timely reporting and preventive measures.
It is crucial that any panic amongst the masses is prevented, he said.
States have been asked to review public health preparedness, particularly at the state and district level by senior officials, besides identification of isolation facilities in hospitals for taking care of both suspected and confirmed cases, availability of logistics and trained human resources and augmentation plan.
“All suspected mpox cases should be isolated and strict infection prevention and control measures put in place. Treatment is symptomatic and available treatment guidelines should be followed,” he said.
Robust diagnostic testing capability is already available, with 36 labs supported by ICMR across the country and three commercial PCR kits validated by ICMR that are now approved by the Central Drugs Standard Control Organisation (CDSCO).
“By implementing the preventive measures and following the recommended management guideline, we can safeguard the health and well-being of individuals and reduce the impact of mpox outbreaks. The Union Ministry of Health shall continue to monitor the situation closely and we will extend all requisite support in this regard to states and Union territories,” the letter said.




